Based on the amplified quenching or superquenching effect, conjugated polyelectrolytes can be used to sense a variety of analytes, such as metal ion, inorganic ions, organic ions, and bio-molecules (DNA, proteins, etc.). For example, the fluorescence of PPE-CO2– can be efficiently quenched by Cu2+. This system was used to sense pyrophosphate, an important anion involved in many biological processes. Because of the stronger binding of Cu2+to PPi, the PPE-CO2– fluorescence is recovered as shown in the following Figure. We have developed a real-time assay for alkaline phosphatase (ALP) based on this system, and efforts to use such a system to sense other important biological analytes, such as ATP, are under the way.

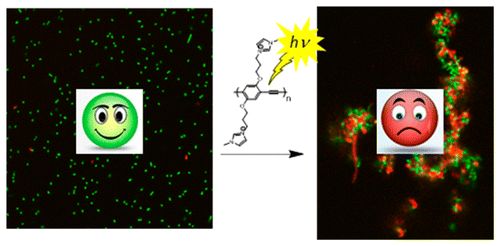
A fluorescence turn-off assay for phospholipase C (PLC) is developed based on the reversible interaction between the natural substrate, phosphatidylcholine and a fluorescent, CPE. The fluorescence intensity of the CPE in water is increased substantially by the addition of the phospholipid due to the formation of a CPE-lipid complex. Incubation of the CPE-lipid complex with the enzyme PLC causes the fluorescence intensity to decrease (turn-off sensor); the response arises due to PLC-catalyzed hydrolysis of the phosphatidylcholine, which effectively disrupts the CPE-lipid complex. The PLC assay operates with phospholipid substrate concentrations in the mM range and the analytical detection limit for PLC is < 1 nM. It demonstrated a good selectivity by testing the response of PLC and other enzymes. The optimized assay provides convenient, rapid and real-time sensor for PLC activity. The real-time fluorescence intensity from the CPE can be converted to substrate concentration by using an ex-situ calibration curve, allowing PLC catalyzed reaction rates and kinetic parameters to be determined.
As another example, meta-inked PPE-type CPEs can self-assemble into a helical conformation in aqueous solution. This allows mPPESO3 to be used as a platform for sensing biological targets with high sensitivity. A biotin-linked Re complex can quench the fluorescence of the polymer. In the presence of avidin, it is expected that due to stronger interaction between biotin and avidin, the intercalation of Re complex into polymer would be disrupted, and the polymer fluorescence would recover.

References:
- Zhao, X.; Liu, Y.; Schanze, K. S. “A Conjugated Polyelectrolyte-based Fluorescence Sensor for Pyrophosphate” Chem. Commun., 2007, 2914-2916.
- Liu, Y.; Ogawa, K.; Schanze, K. S. “Conjugated Polyelectrolyte Based Real-Time Fluorescence Assay for Phospholipase C” Anal. Chem., 2008, 80, 150-158.